At a recent seminar, Dr Xinhua Liu from the Dyson School of Design Engineering discussed her research on Multi-Scale Design for Structural Energy Devices. She has kindly written us this blog to cover the same topics.
What are the bottlenecks to make better batteries?
In a bid to reverse global warming and drastically slash the depletion of fossil fuels and our greenhouse gas pollution, the UK has legally committed itself to reducing 57% of emissions by 2030, and 80% by 2050, these have paved opportunities to worldwide low-carbon drive towards electric vehicles.
But battery technique is frustratingly slow to advance, mainly due to a myriad of reasons:
- Materials selection
- Chemical processes
- Microstructures of electrodes/separator/electrolyte
- Manufacturing process and the resulting single cell performance
- Battery packs that highly rely heavily on good battery management system (BMS) / thermal management system (TMS) designs
Making better batteries is a multiscale and multidisciplinary problem.

Currently lithium ion batteries are the most commercially however there are still many technical challenges (mainly cell-to-cell consistency and thermal uniformity) to overcome. As shown in Figure 1, uneven currents exit in parallel strings due to cell-to-cell variations, thermal gradients and/or cell interconnects which can reduce the overall performance of a battery pack (6P1S).

So it is not fair to extrapolate battery pack performance from single cell performance. We investigated the performance implications caused by these factors by simulating six parallel connected batteries based on a thermally coupled single particle model with the solid electrolyte interphase growth degradation mechanism modelled.
Both model and experimental results show that he placement of the bad cell can affect the output energy. If a higher impedance cell is placed closest to the load point, this has the beneficial impact of equalizing current distributions, however if placed in a location further from the load point this exasperates the problem, leading to reduced performance.
Also, the current heterogeneity can be further influenced by the cell temperature. Therefore, optimizing pack design can achieve higher performance and longer lasting battery packs.
More details can be found in the recent published paper: The effect of cell-to-cell variations and thermal gradients on the performance and degradation of lithium-ion battery packs.
Why we need to improve battery manufacturing techniques?
The ideal batteries should be cheap, high energy & power density, can be safely used in harsh environments, and environmentally friendly. It remains a tremendous challenge for most innovators in battery field to produce new materials at scale which highly rely on developing scalable manufacturing techniques.
Among them, electrospinning has been widely studied in recent years as a low cost, scalable and simple manufacturing technique to produce hierarchical functional nano fibers for various energy storages, delivering a great potential for industrialization of novel materials.
For commercialized batteries, manufacturing techniques are equally critical important to produce cells with the consistent properties and high capacity. For example, the electrode thickness of current commercial cells is about 60-80 µm. If we could make thick electrodes with high energy density batteries, it will largely decrease the cost.
However, the current lithium-ion batteries have one big problem that researchers are dedicating a lot of time to: their cathodes. There are many anode materials that can achieve satisfying capacity, however, the cathode electrodes are greatly limited by their capacity and the mass transfer of thick electrodes. Thus, developing scalable manufacturing techniques for high performance cathode electrode is big challenge, especially the production of thick electrodes with proper microstructure design.
Why design gel electrolyte microstructures?
Most researchers are working on variations of anodes and cathodes, but the electrolyte is also very important. Gel electrolytes are emerging as the main building blocks for energy storage devices (such as lithium ion batteries and supercapacitors) due to their high conductivity and good mechanical flexibility.

In particular, there is a need to develop ionogels with improved thermal and mechanical stability, while maintaining good electrochemical performance through structural alignment. We investigated these microstructures by using XCT and the data were used as the basis for 3D diffusion simulations that proved the enhancement of aligned structure design in electrochemical property. This therefore opens new opportunities for the microstructure design of novel high performance materials for energy storage applications.
How does design move forward structural energy devices?
Structural energy storage devices have the potential to transform products such as aerial vehicles, cars and consumer electronics. 3D printing has developed in recent years, and has been employed to manufacture porous electrodes for lithium-ion batteries, supercapacitors and other related functions.
The electrodes with porous architectures can lead to higher capacities. For example (Figure 3), by using direct metal laser sintering (DMLS), 3D hierarchical scaffolds can be created with high mechanical strength. Functionalisation with MnOx-PEDOT:PSS then imparts the structure with pseduocapacitive properties. Multi-scale x-ray tomography then highlight how this approach improves device performance and lifetime.
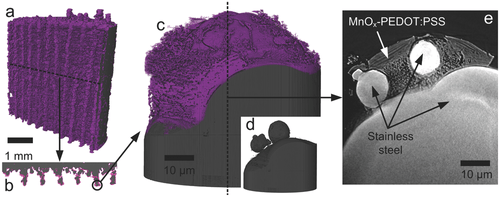
New insights have therefore been presented for the development of 3D printed pseudocapacitors with potential load carrying capabilities, and highlights the opportunity develop other 3D printed energy storage devices.
Are we there yet? Beyond lithium ion battery
Batteries are still stuck, but we are working on it step by step. Dr Billy Wu’s group are currently working on a number of different ways to improve standard battery fabrication techniques. The group is also investigating ways to improve the thermal management systems for battery cells. Their work will allow the degradation of cells, and their potential lifetimes, to be better understood, delivering the great potential to second life batteries. Currently, Dr Chenzhen Ji, Max Naylor-Marlow, Dr Teddy Szemberg O’Connor, Dr Jingyi Chen and myself are working on various projects.
My colleagues and I also focus on promoting scalable electrospinning technology for preparation of energy materials for applications beyond lithium ion battery, such as the functional structural electrodes for solid oxide fuel cell (lead by Dr Mengzheng Ouyang) and free standing film electrodes with hierarchical structure nanofibers for redox flow battery (lead by Dr Barun Chakrabarti).
The on-going H2Go project led by Dr Chenzhen Ji aims to achieve a 3D metal printed H2 storage reactor by proper structure design and integration of heat transfer properties.
There is still some room left to improve existing battery technology, but not enough to meet future requirements. The Electrochemical Science & Engineering (ESE) Group at Imperial College London works at the interface between the science and engineering of electrochemical devices, including lithium-ion batteries, lithium-sulfur batteries, 3D tomography and imaging, redox flow cell, PEM fuel cell, supercapacitors, techno-economic analysis and additive manufacturing.

Dr Xinhua Liu
 Dr Xinhua Liu is a Research Associate at Imperial College London in the Dyson School of Design Engineering and member of the Electrochemical Science and Engineering Group.
Dr Xinhua Liu is a Research Associate at Imperial College London in the Dyson School of Design Engineering and member of the Electrochemical Science and Engineering Group.
She earned her Ph.D. in Material Chemistry and has great interest in electrochemical engineering, working to develop scalable technologies to produce energy materials for various energy applications. With a great interest in design high performance batteries and battery packs from micro-scale to pack level, she has published 30+ papers over the last five years including in Advanced Materials and Advanced Science.
